| << Chapter < Page | Chapter >> Page > |
Finally, the third class of molecules are those that act as intermediaries and can act as both an electron acceptor or as an electron donor, depending upon the situation. Molecules such as NAD + /NADH or cytochrome ox /cytochrome red fall into this category. Think of members of this category as shuttle molecules, shuttling electrons from one compound to another.
How a cell utilizes these three types of molecules is how the cell generates its energy needs, whether it is through ATP generation or by the formation of an "energized membrane" or PMF as we will discuss later.
To understand how red/ox reactions work, we first need to discuss the concept of the half reaction. Two half reactions are required to make the full red/ox reaction. Each half reaction can be thought of as a description of what happens to one of the two molecules involved in the red/ox reaction. This is illustrated in figure 1 below. In this example compound AH is being oxidized by compound B + ; electrons are moving from AH to B + to generate A + BH. Each reaction can be thought of as two coupled half reactions: Where AH is being oxidized and a second reaction where B + is being reduced to BH.
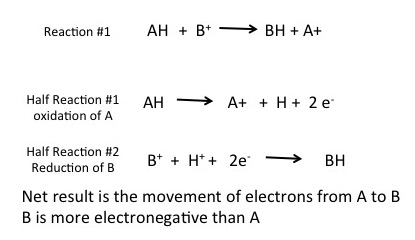
Use figure 1. In reaction #1, AH becomes:
a
By convention we analyze and describe red/ox reactions with respect to reduction potentials that is, with respect to the ability of a compound to gain electrons. Think of this in a similar way to how we think about Acid-Base reactions. By convention we use pH as a measure of the acidity or alkalinity. We view Acid-Base reactions through the lens of acidity. For red/ox reaction we view them through the lens of reduction potentials. That means for every reaction we ask, what is the intrinsic ability of that compound to “attract” or “pull” or “capture” electrons.
What is this intrinsic property to attract electrons? Remember electronegativity , the tendency of an atom or molecule to pull electrons. It is the basis of H-bonding and the ability of molecules to form dipoles or partial charges. This is the same quality or attribute we are discussing in red/ox reactions. Different compounds, based on their structure and composition have intrinsic and distinct attractions for electrons. This quality is termed reduction potential or E 0 ’ and is a relative quantity. Relative in terms of comparison to a “ standard ” reaction. If a compound is more likely to take electrons from the standard, it has a higher or more positive reduction potential. The relative strength of the compound in comparison to the standard can be measured and is given in units of Volts (V) (sometimes written as electron volts or eV) or milliVolts (mV) . So the more positive the reduction potential the stronger the tendency is to take or attract electrons. The more negative the reduction potential the more likely the compound is to get rid of electrons.

Notification Switch
Would you like to follow the 'Ucd bis2a intro to biology v1.2' conversation and receive update notifications?