| << Chapter < Page | Chapter >> Page > |
The dissociation energy of one mole of sodium chloride is therefore
Check Your Understanding If the dissociation energy were larger, would that make it easier or more difficult to break the solid apart?
more difficult
Crystals can also be formed by covalent bonding. For example, covalent bonds are responsible for holding carbon atoms together in diamond crystals. The electron configuration of the carbon atom is —a He core plus four valence electrons. This electron configuration is four electrons short of a full shell, so by sharing these four electrons with other carbon atoms in a covalent bond, the shells of all carbon atoms are filled. Diamond has a more complicated structure than most ionic crystals ( [link] ). Each carbon atom is the center of a regular tetrahedron, and the angle between the bonds is This angle is a direct consequence of the directionality of the p orbitals of carbon atoms.
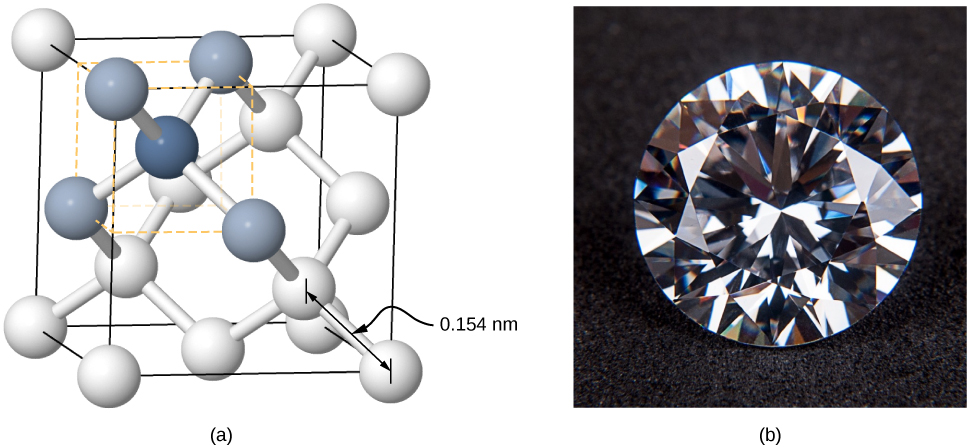
Covalently bonded crystals are not as uniform as ionic crystals but are reasonably hard, difficult to melt, and are insoluble in water. For example, diamond has an extremely high melting temperature (4000 K) and is transparent to visible light. In comparison, covalently bonded tin (also known as alpha-tin, which is nonmetallic) is relatively soft, melts at 600 K, and reflects visible light. Two other important examples of covalently bonded crystals are silicon and germanium. Both of these solids are used extensively in the manufacture of diodes, transistors, and integrated circuits. We will return to these materials later in our discussion of semiconductors.
As the name implies, metallic bonding is responsible for the formation of metallic crystals. The valence electrons are essentially free of the atoms and are able to move relatively easily throughout the metallic crystal. Bonding is due to the attractive forces between the positive ions and the conduction electrons. Metallic bonds are weaker than ionic or covalent bonds, with dissociation energies in the range .
Why is the equilibrium separation distance between different for a diatomic molecule than for solid KCl?
Each ion is in the field of multiple ions of the other opposite charge.
Describe the difference between a face-centered cubic structure (FCC) and a body-centered cubic structure (BCC).
In sodium chloride, how many atoms are “nearest neighbors” of ? How many atoms are “nearest neighbors” of ?
6, 6
In cesium iodide, how many atoms are “nearest neighbors” of ? How many atoms are “nearest neighbors” of ?
The NaCl crystal structure is FCC. The equilibrium spacing is . If each ion occupies a cubic volume of , estimate the distance between “nearest neighbor” ions (center-to-center)?
0.399 nm
The CsI crystal structure is BCC. The equilibrium spacing is approximately . If ion occupies a cubic volume of , what is the distance of this ion to its “nearest neighbor” ion?
0.65 nm
The potential energy of a crystal is /ion pair. Find the dissociation energy for four moles of the crystal.
The measured density of a NaF crystal is . What is the equilibrium separate distance of and ions?
What value of the repulsion constant, n , gives the measured dissociation energy of 221 kcal/mole for NaF?
Determine the dissociation energy of 12 moles of sodium chloride (NaCl). ( Hint: the repulsion constant n is approximately 8.)
2196 kcal
The measured density of a KCl crystal is What is the equilibrium separation distance of and ions?
What value of the repulsion constant, n , gives the measured dissociation energy of 171 kcal/mol for KCl?
11.5
The measured density of a CsCl crystal is . What is the equilibrium separate distance of and ions?

Notification Switch
Would you like to follow the 'University physics volume 3' conversation and receive update notifications?