| << Chapter < Page | Chapter >> Page > |
Note: A voltmeter can also be used in place of the ammeter. A voltmeter will measure the potential difference across the cell.
Results:
During the experiment, you should have noticed the following:
Conclusions:
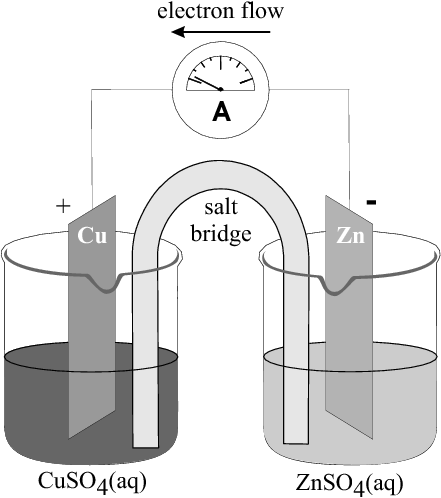
When a zinc sulphate solution containing a zinc plate is connected by a U-tube to a copper sulphate solution containing a copper plate, reactions occur in both solutions. The decrease in mass of the zinc plate suggests that the zinc metal has been oxidised. The increase in mass of the copper plate suggests that reduction has occurred here to produce more copper metal. This will be explained in detail below.
The experiment above demonstrated a zinc-copper cell. This was made up of a zinc half cell and a copper half cell .
A half cell is a structure that consists of a conductive electrode surrounded by a conductive electrolyte. For example, a zinc half cell could consist of a zinc metal plate (the electrode) in a zinc sulphate solution (the electrolyte).
How do we explain what has just been observed in the zinc-copper cell?
In the notation used above, the oxidation half-reaction at the anode is written on the left, and the reduction half-reaction at the cathode is written on the right. In the Zn-Cu electrochemical cell, the direction of current flow in the external circuit is from the zinc electrode (where there has been a build up of electrons) to the copper electrode.
In the zinc-copper cell, the copper and zinc plates are called the electrodes . The electrode where oxidation occurs is called the anode , and the electrode where reduction takes place is called the cathode . In the zinc-copper cell, the zinc plate is the anode and the copper plate is the cathode.

Notification Switch
Would you like to follow the 'Siyavula textbooks: grade 12 physical science' conversation and receive update notifications?