| << Chapter < Page | Chapter >> Page > |
The following video covers some of the properties of an atom.
Veritasium video on the atom - 1
We have now looked at many examples of the types of matter and materials that exist around us and we have investigated some of the ways that materials are classified. But what is it that makes up these materials? And what makes one material different from another? In order to understand this, we need to take a closer look at the building block of matter - the atom . Atoms are the basis of all the structures and organisms in the universe. The planets, sun, grass, trees, air we breathe and people are all made up of different combinations of atoms.
It is important to realise that a lot of what we know about the structure of atoms has been developed over a long period of time. This is often how scientific knowledge develops, with one person building on the ideas of someone else. We are going to look at how our modern understanding of the atom has evolved over time.
The idea of atoms was invented by two Greek philosophers, Democritus and Leucippus in the fifth century BC. The Greek word
Nowadays, we know that atoms are made up of a positively charged nucleus in the centre surrounded by negatively charged electrons . However, in the past, before the structure of the atom was properly understood, scientists came up with lots of different models or pictures to describe what atoms look like.
A model is a representation of a system in the real world. Models help us to understand systems and their properties. For example, an atomic model represents what the structure of an atom could look like, based on what we know about how atoms behave. It is not necessarily a true picture of the exact structure of an atom.
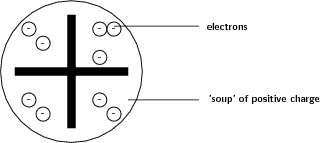
After the electron was discovered by J.J. Thomson in 1897, people realised that atoms were made up of even smaller particles than they had previously thought. However, the atomic nucleus had not been discovered yet and so the 'plum pudding model' was put forward in 1904. In this model, the atom is made up of negative electrons that float in a soup of positive charge, much like plums in a pudding or raisins in a fruit cake ( [link] ). In 1906, Thomson was awarded the Nobel Prize for his work in this field. However, even with the Plum Pudding Model, there was still no understanding of how these electrons in the atom were arranged.
The discovery of radiation was the next step along the path to building an accurate picture of atomic structure. In the early twentieth century, Marie Curie and her husband Pierre, discovered that some elements (the radioactive elements) emit particles, which are able to pass through matter in a similar way to X-rays (read more about this in Grade 11). It was Ernest Rutherford who, in 1911, used this discovery to revise the model of the atom.

Notification Switch
Would you like to follow the 'Siyavula textbooks: grade 10 physical science' conversation and receive update notifications?