| << Chapter < Page | Chapter >> Page > |
The rates at which reactants are consumed and products are formed during chemical reactions vary greatly. We can identify five factors that affect the rates of chemical reactions: the chemical nature of the reacting substances, the state of subdivision (one large lump versus many small particles) of the reactants, the temperature of the reactants, the concentration of the reactants, and the presence of a catalyst.
The rate of a reaction depends on the nature of the participating substances. Reactions that appear similar may have different rates under the same conditions, depending on the identity of the reactants. For example, when small pieces of the metals iron and sodium are exposed to air, the sodium reacts completely with air overnight, whereas the iron is barely affected. The active metals calcium and sodium both react with water to form hydrogen gas and a base. Yet calcium reacts at a moderate rate, whereas sodium reacts so rapidly that the reaction is almost explosive.
Except for substances in the gaseous state or in solution, reactions occur at the boundary, or interface, between two phases. Hence, the rate of a reaction between two phases depends to a great extent on the surface contact between them. A finely divided solid has more surface area available for reaction than does one large piece of the same substance. Thus a liquid will react more rapidly with a finely divided solid than with a large piece of the same solid. For example, large pieces of iron react slowly with acids; finely divided iron reacts much more rapidly ( [link] ). Large pieces of wood smolder, smaller pieces burn rapidly, and saw dust burns explosively.
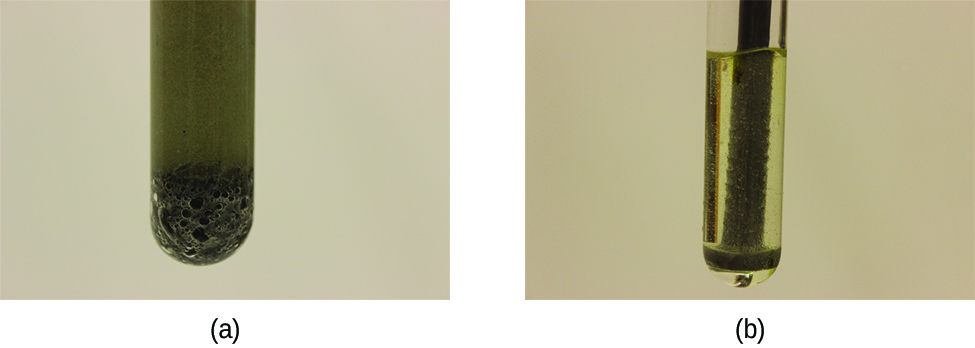
Watch this video to see the reaction of cesium with water in slow motion and a discussion of how the state of reactants and particle size affect reaction rates.
Chemical reactions typically occur faster at higher temperatures. Food can spoil quickly when left on the kitchen counter. However, the lower temperature inside of a refrigerator slows that process so that the same food remains fresh for days. We use a burner or a hot plate in the laboratory to increase the speed of reactions that proceed slowly at ordinary temperatures. In many cases, an increase in temperature of only 10 °C will approximately double the rate of a reaction in a homogeneous system.
The rates of many reactions depend on the concentrations of the reactants. Rates usually increase when the concentration of one or more of the reactants increases. For example, calcium carbonate (CaCO 3 ) deteriorates as a result of its reaction with the pollutant sulfur dioxide. The rate of this reaction depends on the amount of sulfur dioxide in the air ( [link] ). An acidic oxide, sulfur dioxide combines with water vapor in the air to produce sulfurous acid in the following reaction:

Notification Switch
Would you like to follow the 'Ut austin - principles of chemistry' conversation and receive update notifications?