| << Chapter < Page | Chapter >> Page > |
At the heart of ATP is a molecule of adenosine monophosphate (AMP), which is composed of an adenine molecule bonded to a ribose molecule and to a single phosphate group ( [link] ). Ribose is a five-carbon sugar found in RNA, and AMP is one of the nucleotides in RNA. The addition of a second phosphate group by a condensation reaction to this core molecule results in the formation of adenosine di phosphate (ADP); the addition of a third phosphate group by a condensation reaction forms adenosine tri phosphate (ATP).
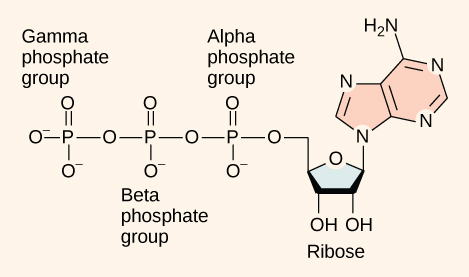
The addition of a phosphate group to a molecule requires energy. Phosphate groups are negatively charged and thus repel one another when they are arranged in series, as they are in ADP and ATP. This repulsion makes the ADP and ATP molecules inherently unstable. The release of one or two phosphate groups from ATP, a process called hydrolysis , releases energy.
Hydrolysis is the process of breaking complex macromolecules apart. During hydrolysis, water is split, or lysed, and the resulting hydrogen atom (H
+ ) and a hydroxyl group (OH
- ) are added to the larger molecule. The hydrolysis of ATP produces ADP, together with an inorganic phosphate ion (P
i ), and the release of free energy an exergonic reaction. This free energy is then available to drive an endergonic reaction. The release of energy and absorption of this free energy is a
coupled reaction . To carry out life processes, ATP is continuously broken down into ADP, and like a rechargeable battery, ADP is continuously regenerated into ATP by the reattachment of a third phosphate group by a condensation reaction (
[link] ). Water, which was broken down into its hydrogen atom and hydroxyl group during ATP hydrolysis, is reformed when a third phosphate is added to the ADP molecule, reforming ATP with energy being absorbed. The energy necessary for driving the formation of ATP comes from an exergonic reaction. Once again the coupling of an exergonic reaction drives an endergonic reaction or the formation of ATP.
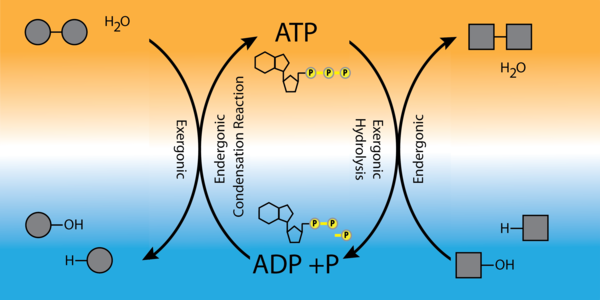
Obviously, energy must be infused into the system to regenerate ATP. Where does this energy come from? In nearly every living thing on earth, the energy comes from the metabolism of glucose. In this way, ATP is a direct link between the limited set of exergonic pathways of glucose catabolism and the multitude of endergonic pathways that power living cells.
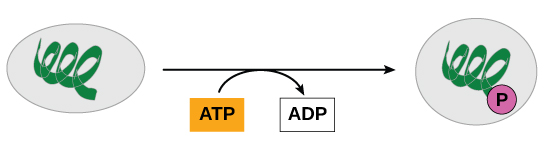
Recall that, in some chemical reactions, enzymes may bind to several substrates that react with each other on the enzyme, forming an intermediate complex. An intermediate complex is a temporary structure, and it allows one of the substrates (such as ATP) and reactants to more readily react with each other; in reactions involving ATP, ATP is one of the substrates and ADP is a product. During an endergonic chemical reaction, ATP forms an intermediate complex with the substrate and enzyme in the reaction. This intermediate complex allows the ATP to transfer its third phosphate group, with its energy, to the substrate, a process called phosphorylation. Phosphorylation refers to the addition of the phosphate (~P). This is illustrated by the following generic reaction:
When the intermediate complex breaks apart, the energy is used to modify the substrate and convert it into a product of the reaction. The ADP molecule and a free phosphate ion are released into the medium and are available for recycling through cell metabolism.
ATP is generated through two mechanisms during the breakdown of glucose. A few ATP molecules are generated (that is, regenerated from ADP) as a direct result of the chemical reactions that occur in the catabolic pathways. A phosphate group is removed from an intermediate reactant in the pathway, and the free energy of the reaction is used to add the third phosphate to an available ADP molecule, producing ATP ( [link] ). This very direct method of phosphorylation is called substrate-level phosphorylation .
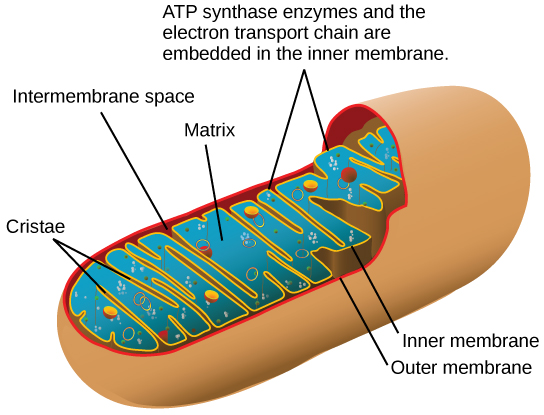
Most of the ATP generated during glucose catabolism, however, is derived from a much more complex process, chemiosmosis, which takes place in mitochondria ( [link] ) within a eukaryotic cell or the plasma membrane of a prokaryotic cell. Chemiosmosis , a process of ATP production in cellular metabolism, is used to generate 90 percent of the ATP made during glucose catabolism and is also the method used in the light reactions of photosynthesis to harness the energy of sunlight. The production of ATP using the process of chemiosmosis is called oxidative phosphorylation because of the involvement of oxygen in the process.

Notification Switch
Would you like to follow the 'Principles of biology' conversation and receive update notifications?