| << Chapter < Page | Chapter >> Page > |
Relative atomic mass is the average mass of one atom of all the naturally occurring isotopes of a particular chemical element, expressed in atomic mass units.
The relative atomic mass of some elements depends on where on Earth the element is found. This is because the isotopes can be found in varying ratios depending on certain factors such as geological composition, etc. The International Union of Pure and Applied Chemistry (IUPAC) has decided to give the relative atomic mass of some elements as a range to better represent the varying isotope ratios on the Earth. For the calculations that you will do at high school, it is enough to simply use one number without worrying about these ranges.
The element chlorine has two isotopes, chlorine-35 and chlorine-37. The abundance of these isotopes when they occur naturally is 75% chlorine-35 and 25% chlorine-37. Calculate the average relative atomic mass for chlorine.
Contribution of
Contribution of
If you look on the periodic table, the average relative atomic mass for chlorine is . You will notice that for many elements, the relative atomic mass that is shown is not a whole number. You should now understand that this number is the average relative atomic mass for those elements that have naturally occurring isotopes.
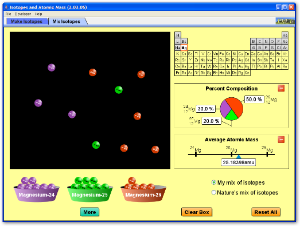
This simulation allows you to see how isotopes and relative atomic mass are inter related.
| Isotope | Z | A | Protons | Neutrons | Electrons |
| Carbon-12 | |||||
| Carbon-14 | |||||
| Chlorine-35 | |||||
| Chlorine-37 |
Scientific knowledge is not static: it changes and evolves over time as scientists build on the ideas of others to come up with revised (and often improved) theories and ideas. In this chapter for example, we saw how peoples' understanding of atomic structure changed as more information was gathered about the atom. There are many more examples like this one in the field of science. For example, think about our knowledge of the solar system and the origin of the universe, or about the particle and wave nature of light.
Often, these changes in scientific thinking can be very controversial because they disturb what people have come to know and accept. It is important that we realise that what we know now about science may also change. An important part of being a scientist is to be a critical thinker . This means that you need to question information that you are given and decide whether it is accurate and whether it can be accepted as true. At the same time, you need to learn to be open to new ideas and not to become stuck in what you believe is right... there might just be something new waiting around the corner that you have not thought about!

Notification Switch
Would you like to follow the 'Chemistry grade 10 [caps]' conversation and receive update notifications?