| << Chapter < Page | Chapter >> Page > |
The earth's atmosphere is made up of about 78% nitrogen, making it the largest pool of this gas. Nitrogen is essential for many biological processes. It is in all amino acids, proteins and nucleic acids. As you will see in a later chapter, these compounds are needed to build tissues, transport substances around the body and control what happens in living organisms. In plants, much of the nitrogen is used in chlorophyll molecules which are needed for photosynthesis and growth.
So, if nitrogen is so essential for life, how does it go from being a gas in the atmosphere to being part of living organisms such as plants and animals? The problem with nitrogen is that it is an 'inert' gas, which means that it is unavailable to living organisms in its gaseous form. This is because of the strong triple bond between its atoms that makes it difficult to break. Something needs to happen to the nitrogen gas to change it into a form that it can be used. And at some later stage, these new compounds must be converted back into nitrogen gas so that the amount of nitrogen in the atmosphere stays the same. This process of changing nitrogen into different forms is called the nitrogen cycle ( [link] ).
The nitrogen cycle is a biogeochemical cycle that describes how nitrogen and nitrogen-containing compounds are changed in nature.
Very broadly, the nitrogen cycle is made up of the following processes:
We are going to look at each of these processes in more detail.
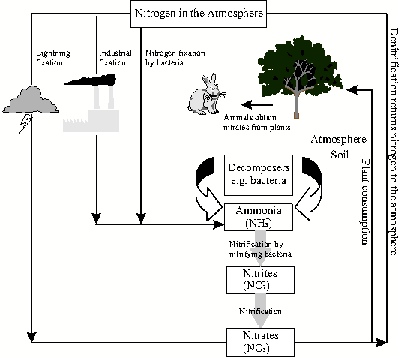
Nitrogen fixation is needed to change gaseous nitrogen into forms such as ammonia that are more useful to living organisms. Some fixation occurs in lightning strikes and in industrial processes, but most fixation is done by different types of bacteria living either in the soil or in parts of the plants.
It is interesting to note that by cultivating legumes, using the Haber-Bosch process to manufacture chemical fertilisers and increasing pollution from vehicles and industry, humans have more than doubled the amount of nitrogen that would normally be changed from nitrogen gas into a biologically useful form. This has serious environmental consequences.
Nitrification involves two biological oxidation reactions: firstly, the oxidation of ammonia with oxygen to form nitrite (NO 2 ) and secondly the oxidation of these nitrites into nitrates.
Nitrification is an important step in the nitrogen cycle in soil because it converts the ammonia (from the nitrogen fixing part of the cycle) into nitrates, which are easily absorbed by the roots of plants. This absorption of nitrates by plants is called assimilation . Once the nitrates have been assimilated by the plants, they become part of the plants' proteins. These plant proteins are then available to be eaten by animals. In other words, animals (including humans) obtain their own nitrogen by feeding on plants. Nitrification is performed by bacteria in the soil, called nitrifying bacteria .
Read the information below and then carry out your own research to help you answer the questions that follow.
The negatively charged nitrate ion is not held onto soil particles and so can be easily washed out of the soil. This is called leaching . In this way, valuable nitrogen can be lost from the soil, reducing the soil's fertility. The nitrates can then accumulate in groundwater and eventually in drinking water. There are strict regulations that control how much nitrate can be present in drinking water, because nitrates can be reduced to highly reactive nitrites by microorganisms in the gut. Nitrites are absorbed from the gut and bind to haemoglobin (the pigment in blood that helps to transport oxygen around the body). This reduces the ability of the haemoglobin to carry oxygen. In young babies this can lead to respiratory distress, a condition known as "blue baby syndrome".
Denitrification is the process of reducing nitrate and nitrite into gaseous nitrogen. The process is carried out by denitrification bacteria . The nitrogen that is produced is returned to the atmosphere to complete the nitrogen cycle.
The equation for the reaction is:
2NO 3 + 10e + 12H + N 2 + 6H 2 O

Notification Switch
Would you like to follow the 'Siyavula textbooks: grade 10 physical science' conversation and receive update notifications?