| << Chapter < Page | Chapter >> Page > |
| Chemical Reactions of Hydrogen with Other Elements | |
|---|---|
| General Equation | Comments |
| ionic hydrides with group 1 and Ca, Sr, and Ba | |
| requires high pressure and temperature; low yield | |
| exothermic and potentially explosive | |
| requires heating; low yield | |
| X = F, Cl, Br, and I; explosive with F 2 ; low yield with I 2 |
Hydrogen reduces the heated oxides of many metals, with the formation of the metal and water vapor. For example, passing hydrogen over heated CuO forms copper and water.
Hydrogen may also reduce the metal ions in some metal oxides to lower oxidation states:
Other than the noble gases, each of the nonmetals forms compounds with hydrogen. For brevity, we will discuss only a few hydrogen compounds of the nonmetals here.
Ammonia, NH 3 , forms naturally when any nitrogen-containing organic material decomposes in the absence of air. The laboratory preparation of ammonia is by the reaction of an ammonium salt with a strong base such as sodium hydroxide. The acid-base reaction with the weakly acidic ammonium ion gives ammonia, illustrated in [link] . Ammonia also forms when ionic nitrides react with water. The nitride ion is a much stronger base than the hydroxide ion:
The commercial production of ammonia is by the direct combination of the elements in the Haber process :
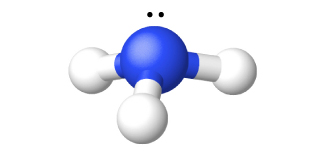
Ammonia is a colorless gas with a sharp, pungent odor. Smelling salts utilize this powerful odor. Gaseous ammonia readily liquefies to give a colorless liquid that boils at −33 °C. Due to intermolecular hydrogen bonding, the enthalpy of vaporization of liquid ammonia is higher than that of any other liquid except water, so ammonia is useful as a refrigerant. Ammonia is quite soluble in water (658 L at STP dissolves in 1 L H 2 O).
The chemical properties of ammonia are as follows:

Notification Switch
Would you like to follow the 'Chemistry' conversation and receive update notifications?